Lungs Betray the Brain: Nicotine Hijacks Rare Lung Cells to Ship Iron “Packages” That Trigger Dementia-Linked Damage in Neurons
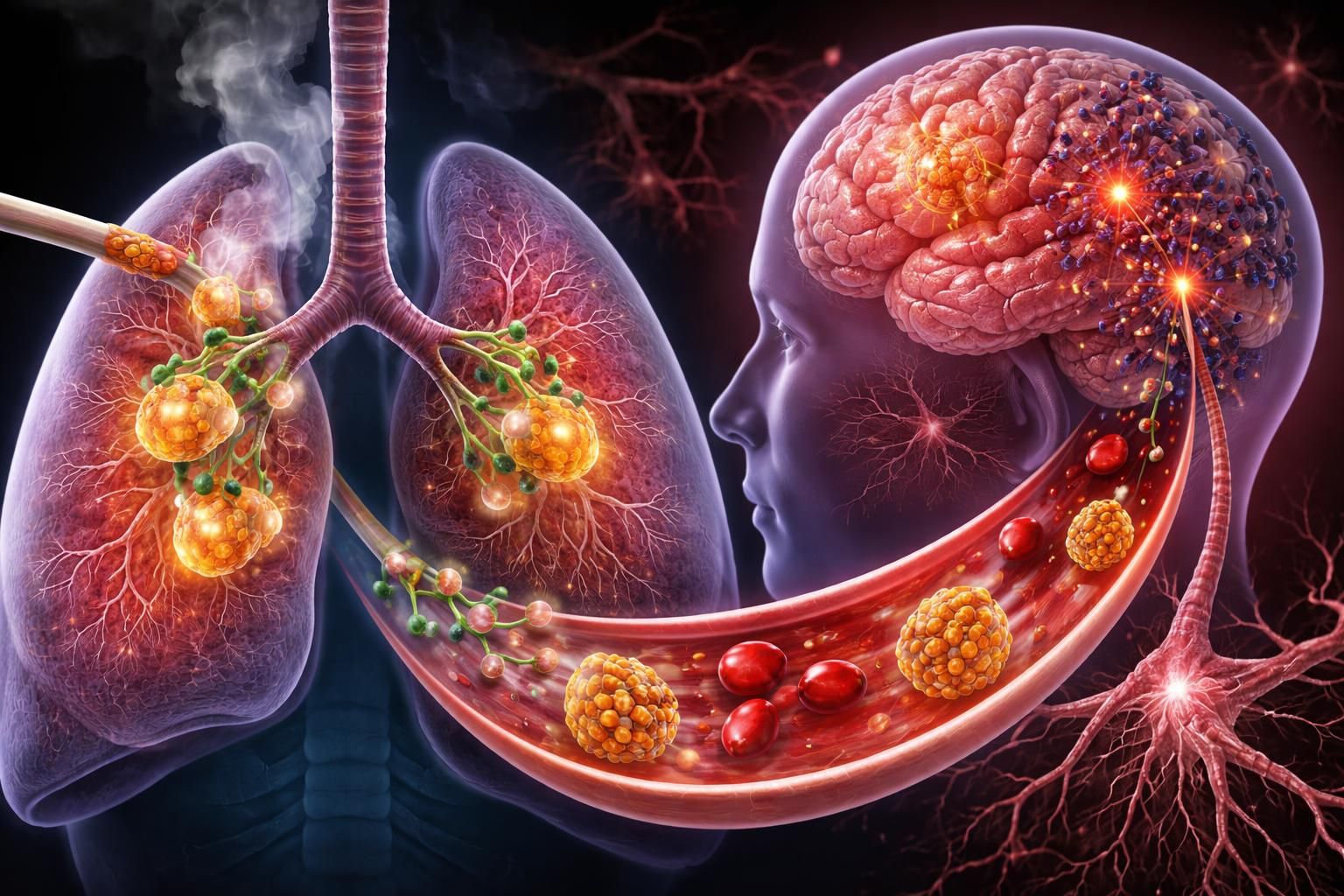
A groundbreaking study from the University of Chicago has uncovered a previously unknown biological pathway that may help explain why smoking is strongly linked to higher risks of dementia and neurodegenerative diseases. Led by Asst. Prof. Huanhuan Joyce Chen of the Pritzker School of Molecular Engineering and the Ben May Department for Cancer Research, the research reveals how nicotine prompts specialized lung cells to release tiny vesicles packed with iron-regulating proteins. These “exosomes” travel to nearby neurons, disrupt iron balance, and spark cellular stress resembling early stages of Parkinson’s and Alzheimer’s disease.
The overlooked “lung-brain axis”
Smoking has long been associated with cognitive decline—epidemiological data show heavy midlife smoking can more than double the risk of Alzheimer’s and other dementias later in life. Traditional explanations focused on vascular damage, reduced oxygen delivery, or direct nicotine effects on the brain. This new work adds a surprising twist: the lungs are not just passive victims of smoke but active participants that send harmful signals directly to the nervous system. At the center of the discovery are pulmonary neuroendocrine cells (PNECs)—rare, sensory-like cells that make up less than 1% of all lung cells. Because they are so scarce, studying them directly has been nearly impossible. The Chicago team solved this by generating “induced PNECs” (iPNECs) from human pluripotent stem cells, creating enough material for rigorous experiments.
Nicotine flips the switch
When these iPNECs were exposed to nicotine (the addictive component of cigarettes and vapes), they dramatically ramped up exosome production—releasing nearly 2.8 times more of these microscopic packages than untreated cells. The exosomes were not ordinary; they were heavily loaded with serotransferrin, the body’s primary iron-transport protein. Neurons (specifically human dorsal-root-ganglion-like sensory neurons that mirror those connected to the lungs via the vagus nerve) readily absorbed these nicotine-triggered exosomes. Inside the neurons, the extra serotransferrin drove iron overload: levels of key iron-import proteins (TFR1, DMT1, and DCYTB) rose, ferritin (the iron-storage protein) accumulated, mitochondria lost function, energy (ATP) production dropped, and harmful reactive oxygen species surged. Most tellingly, the neurons began producing higher levels of α-synuclein—the protein that clumps in Parkinson’s disease and is linked to broader neurodegeneration. The researchers also showed the pathway is reversible in the lab. Blocking exosome release with the inhibitor GW4869, silencing the transferrin gene in PNECs, or inhibiting the transferrin receptor on neurons all reduced or prevented the iron imbalance, oxidative stress, and α-synuclein spike.
What this means for human health
The study proposes a direct “lung-brain axis” in which PNECs, acting as airway sensors, use exosomes to communicate with neighboring sensory neurons. Nicotine hijacks this normal signaling route, turning a protective or homeostatic mechanism into a source of chronic neuronal stress. Because the vagus nerve connects lungs to brain, these local changes could propagate farther, contributing to the cognitive risks long observed in smokers. Co-first author Kui Zhang, a postdoctoral researcher, emphasized: “This research establishes a clear ‘lung-brain’ axis that helps explain why cigarette smoking is linked to cognitive decline and neurodegenerative risks. By understanding how these exosomes perturb iron homeostasis, we open new doors for protecting neurons from smoke-induced damage.” Corresponding author Joyce Chen added: “It reveals that the lung is not just a passive target of smoke exposure, but an active signaling organ influencing brain pathology… Understanding these cross-organ communication pathways is critical for developing better prevention and intervention strategies for neurodegenerative diseases.”
Important caveats
The findings are compelling but preliminary. Most experiments were conducted in human stem-cell-derived models and ex-vivo mouse lung tissue; direct proof in living animals or long-term human studies is still needed. The authors stress that future animal models will be essential to confirm whether this pathway actually drives dementia risk in real-world smoking scenarios and whether blocking the exosome–iron route could become a therapeutic target.
Citation
Thakur, A., Zhang, K., Chen, J., Mei, S., Chen, J., Verdiell, A., Jacobs, E., Mulder, L., Whitford, A., Iyaswamy, A., Chen, Y.-W., Cliffe, A., Esser-Kahn, A., Chen, B., Lian, Q., Solway, J., Xing, S., & Chen, H. J. (2026). Pulmonary neuroendocrine cell–derived exosomes regulate iron homeostasis and oxidative stress in lung neurons. Science Advances, 12(15), eady2696. https://doi.org/10.1126/sciadv.ady2696




Leave a Reply